FINAL REPORT:
Intertidal Lobster Monitoring Program:
1993-1999
Submitted to the
Maine Department of Marine Resources
in partial fulfillment of Contract # 99872
by
Diane F. Cowan, Ph.D., and Sara L. Ellis, Ph.D.
March 30, 2000
Intertidal Lobster Monitoring Program, 1993-1999
The primary objective of The Lobster Conservancy’s Intertidal Lobster Monitoring Program (ILMP) is to develop a method for assessing the abundance and distribution of young-of-the-year and early juvenile lobsters. The implementation of a long-term monitoring program allows us to assess the health and productivity of lobster nurseries in much the same way that clam flats and water quality have been studied. Initially, the focus of this study has been on Casco Bay, which has served as a testing ground and model study for intertidal areas identified as lobster nurseries throughout New England.
The ILMP presents an opportunity to continue monitoring the abundance of juvenile lobster in a quantitative manner that assesses population size, postlarval settlement, and juvenile growth. By measuring the number of lobsters per unit area month after month and year after year, The Lobster Conservancy is developing a time series of baseline information on population trends of juvenile lobsters. In addition to estimating population size, biologists need to know (1) the time lag between infancy and adulthood (growth rate) and (2) the relative number of individuals reaching adulthood (mortality rate). To generate the data required to estimate rates of growth and mortality, The Lobster Conservancy (TLC) has implemented a tag-and-release program. In this study, each lobster is marked by implanting an individually coded tag into the muscle tissue. Taken together these data may be used to predict or forecast the future health of the lobster industry by foreseeing changes in stock abundance.
Tagging Rationale
Lobsters grow by shedding their shell periodically in a process known as molting. They spend all of their lives preparing for, recovering from, and actually molting (except for the 9-11 month break females take for carrying eggs). Molting is the only way a lobster can grow because the shell is a non-living inelastic exoskeleton — unlike our living bone that grows within us. The fact that lobsters molt has posed a problem for scientists who want to know how long it takes them to grow up because any external marker that could be placed on a lobster is lost when it sheds its shell. New technology has made it possible to tag lobsters internally and track them over time — even through their molts.
The Lobster Conservancy has taken advantage of this method by tagging approximately 9,000 lobsters since September 1993 (Cowan 1999a, Cowan 1999b). The data gathered on these lobsters offer the first clues as to (1) how quickly lobsters grow in nature, (2) how old lobsters are at a given size, (3) how faithful lobsters are to their home cove, and (4) what time of year and at what age lobsters are making small- and large-scale movements.
Methods
Quadrat Sampling
The sentinel site for TLC’s long-term monitoring program is Lowell's Cove, on Orr's Island in Casco Bay, Maine. Dr. Diane F. Cowan began year-round surveys at Lowell’s Cove in February 1993, using a standard ecological method for estimating abundance of organisms (Cowan 1999a, 1999b). Surveys were conducted by sampling all lobsters found in 1-m2 quadrats along fixed horizontal transects during the 14 weeks of the lowest tides of each year (i.e., during all tides that were predicted to fall a minimum of 0.2 m below mean low water). To maximize the time when lobsters were exposed, quadrat sampling began approximately 1 h before the predicted time of low water and continued for at least 1 h thereafter.
Monthly quadrat sampling was conducted when tides ranged from -0.2 to -0.6 m. The area sampled each month varied with lobster abundance, due to time constraints of measuring and tagging individuals in the field. Air, water, and substrate temperatures were recorded to the nearest 0.1°C using a Hannah instruments H19063 microcomputer K-thermocoupled thermometer at the onset and end of each sampling period. Surface salinity of the cove was measured at the same time, using a temperature-compensated handheld salinity refractometer (American Optical Corporation).
A qualitative description of each quadrat was made by estimating the percentage of rock cover and substrate type, the presence of each macroalgal and animal species observed, and the presence or absence of depressions filled with water. Movable rocks in the quadrats were then overturned one at a time and organisms found beneath the rocks were recorded.
When juvenile lobsters were found, the following information was recorded: carapace length - (from the rear of the eye socket to the posterior margin of the carapace), total length (from the tip of the rostrum to the tip of the telson), handedness (right or left crusher), sex (for lobsters measuring >15 mm carapace length), condition of appendages (i.e., missing, regenerating, damaged), molt condition (appearance of exoskeleton and limb buds; Waddy et al, 1995), condition of tissues (i.e., evidence of swelling at low salinities), and activity (any movement of appendages or entire body). Carapace length was measured to the nearest 0.5 mm using vernier calipers and total length to the nearest 1 mm using a hand ruler.
Tagging
To mark juvenile lobsters, a tiny (1-mm long x 0.25-mm diameter), individually coded, magnetized microwire tag (Northwest Marine Technology, Shaw Island, WA) was injected directly into the muscle tissue of the second walking leg where the thumb opens up to expose a fleshy part (method modified for marking lobsters by Krouse and Nutting 1990). The tag was injected using a hypodermic needle with a wire plunger that pokes into the lobster through the syringe, thereby pushing the tag into the muscle tissue.
When a tagged lobster was recaptured, the tag was discovered by a high-pitched beep emitted by a scanning detector, signifying that the lobster has been previously tagged. The tag was removed, and the lobster was retagged and released. The original tag was brought back to the lab, where a series of bar codes on each tag was carefully deciphered under a dissecting microscope to determine the identity of the individual. This identification process, combined with information gathered during quadrat sampling, allows us to gather data on growth rates and movement patterns of individual lobsters.
Lobsters that were found during the course of quadrat sampling were tagged. In addition, lobsters were gathered through haphazard sampling higher in the intertidal zone, in tide pools, and in other regions of the cove in an attempt to recapture tagged lobsters. In most cases, only lobsters measuring more than 15 mm carapace length were tagged. Lobsters could not be tagged when the air temperature fell below 5°C due to their propensity for autotomizing limbs at cold temperatures.
All tagging data were recorded on Sony Microcassette™ 3MC-60 audiocassette tapes. Tapes were later transcribed onto data sheets and information was entered into an Excel database. Recapture analyses were done using SYSTAT. Means are presented ± standard error.
Volunteer Monitoring
Starting in 1997, the ILMP was extended beyond Lowell’s Cove with the assistance of volunteer monitors. Volunteers sample specific sites once per month during the spring low tides from May through October. As of October 1999, TLC had 43 volunteers monitoring 23 sites in Maine and 1 site in New Hampshire (Figure 1). Monitors conducted quadrat sampling along a 20-meter transect, and collected the same data on lobster characteristics and environmental parameters as described above. Volunteers did not, however, conduct tagging studies.
Results
Between September 1993 and November 1999, 8,509 lobsters were tagged in situ at Lowell's Cove. Carapace length of tagged lobsters ranged from 10 to 83 mm, with a mean of 30.0 ± 0.09 mm.
Among the tagged lobsters, sex ratio was skewed heavily toward males, with a sex ratio of 2:1 (Table 1). The male-biased sex ratio is strikingly consistent at all intertidal sites throughout Harpswell, Penobscot Bay, and New Hampshire (Table 1). The reasons for the skewed sex ratio in the intertidal zone are not known. In lobsters, sex is not determined until several molts after settlement. It is possible that environmental factors such as temperature or salinity in the intertidal zone lead to the development of more males than females. Alternatively, the male-biased sex ratio may reflect differential rates of survival in, or tolerance to, this environment. Long-term analyses of juvenile lobsters in the subtidal zone suggest that sex ratios in this deeper area are closer to 1:1 (Steneck, unpublished).
We measured the incidence of injury by recording claw loss. At Lowell’s Cove, 76% of lobsters had both claws. Only 4% of tagged lobsters were missing both claws, while 18% were missing one claw (Table 2). Similar patterns of injury were observed at other intertidal sites, where the vast majority of lobsters had both claws (Table 2).
Table 1. Sex ratio and claw loss of juvenile lobsters at The Lobster Conservancy's intertidal study sites around New England.
| Area |
Male : Female
|
Sample size
|
| Lowell's Cove |
2.0 : 1 |
8,513 |
| Penobscot Bay1 |
2.0 : 1 |
335 |
| Harpswell2 |
1.6 : 1 |
382 |
| New Hampshire3 |
2.0 : 1 |
214 |
1 Ellis and Cowan 1999a; 2 Ellis and Cowan 1999b; 3Ellis and Cowan 1999c
Table 2. Injury rate of juvenile lobsters at The Lobster Conservancy's intertidal study sites around New England.
| Area |
No missing claws
(%)
|
1 missing claw
(%)
|
2 missing claws
(%)
|
Sample size
|
| Lowell's Cove |
78 |
18 |
4 |
8,509 |
| Penobscot Bay1 |
76 |
18 |
6 |
335 |
| Harpswell2 |
76 |
20 |
4 |
358 |
| New Hampshire3 |
65 |
25 |
10 |
214 |
1 Ellis and Cowan 1999a; 2 Ellis and Cowan 1999b; 3Ellis and Cowan 1999c
Tag and Recapture
Of the tagged lobsters, 821 were recaptured, yielding an overall recapture rate of 9.6% (Table 3). Although most recaptured individuals were caught only twice, 7% percent were captured 3 or more times (Table 4). Many of the recaptured lobsters had molted at least once since the time of first capture as evidenced by incremental changes in length (Figure 2). The change in carapace length tends to be greater for larger individuals (Figure 2). The high degree of variability in the data is largely due to differences in the time interval between first and subsequent capture.
Table 3. Rates of tagging and recapture of juvenile lobsters at Lowell's Cove, Casco Bay, Maine using binary-coded microwire tags (n = 8,509).
| Year |
Number tagged
|
Number recaptured
|
Recapture rate (%)
|
| 1999 |
1,149
|
153
|
13.3 |
| 1998 |
1,815
|
221
|
12.2 |
| 1997 |
1,463
|
184
|
12.6 |
| 1996 |
1,748
|
128
|
7.3 |
| 1995 |
1,184
|
74
|
6.3 |
| 1994 |
933
|
52
|
5.6 |
| 1993 |
217
|
9
|
4.1 |
| Overall |
8,509
|
821
|
9.6 |
Table 4. Recaptured lobsters were caught between 2 and 6 times at Lowell's Cove, Casco Bay, Maine (n = 728).
|
Times captured
|
Number of lobsters
|
Percentage of total
|
|
2
|
679
|
93.3
|
|
3
|
39
|
5.4
|
|
4
|
7
|
1.0
|
|
5
|
2
|
0.3
|
|
6
|
1
|
0.1
|
The time interval between first and second capture varied from 30 to 900 days and the change in carapace length varied from 0 to 22 mm over the same period of time (Figure 3). As expected, change in carapace length increased with number of days at large. The greatest changes in carapace length were recorded for individuals who escaped recapture for the longest duration.
The following case study of 6 lobsters captured on 4 or more occasions shows the kind of details that are available through multiple tag and recapture studies (Figure 4). Female lobster #2 was captured on five separate occasions (Figure 4). At the time of initial capture on 23 July 1994, lobster #2 measured 18 mm CL. She was next captured on 14 June 1995 at a new size of 23 mm CL. During the summer of 1995, female #2 underwent two additional molts, growing to 27.5 mm CL by 12 July 12 1995, and to 31 mm CL by 9 September 1995. Each time female #2 was captured in 1994 and 1995, she was within seven meters of the point of initial capture. Female #2 was not captured again until 6 June 1996, after 270 days at large. She was found approximately 50 meters away, but had not molted. Individuals 2, 4, and 6 were each captured at 30-day intervals in the summer months. Their incremental changes in carapace length over that time period most likely represent the growth increase of one molting event. The long periods of time lobsters 1, 2, and 3 did not grow in over 200 days at large represent winter months. Overall, there appears to be a pattern for individuals of these sizes to molt two to three times per year, during the summer months.
Patterns of Abundance
We observed a seasonal pattern of lobster abundance in the intertidal zone. The highest densities were recorded at Lowell’s Cove from May through November (Figure 5). Lower densities in December through April were most likely due to seasonal migrations of yearlings and older juveniles to deeper water since tagging studies showed that such individuals disappeared and reappeared abruptly.
To examine interannual variation in abundance, we compared lobster density at Lowell’s Cove during the peak months for each year (September-November; Figure 6). The abundance of lobsters of all size classes increased from 1993-1995. Overall densities, and densities of young-of the-year (YOY) peaked in 1995, the warmest year since sampling began. The decrease in YOY density in 1996 was followed by a decrease in the larger size class with a one-year time lag. Density of YOY decreased further in 1997 and remained at this lower level in 1998 (Figure 6).
Intertidal versus Subtidal Abundance
To compare abundance estimates gathered by the Intertidal Lobster Monitoring Program with estimates based on data gathered by suction sampling subtidally (Steneck, unpublished), we compared fall densities at 13 sites in Penobscot Bay, Maine (Figure 7). There was a strong positive correlation in densities of Early Benthic Phase lobsters (< 40 mm CL) (r = 0.87, p < 0.001).
Discussion
Significance of lobsters’ use of the intertidal zone
Throughout its range, cover is a critical requirement for shelter-dependent juvenile lobsters that find refuge in various substrates including cobble, rock and boulder (Bernstein and Campbell, 1983; Hudon, 1987; Barshaw and Bryant-Rich, 1988; Barshaw and Lavalli, 1988; Wahle and Steneck, 1991, 1992), eel grass (Barshaw and Lavalli, 1988; Heck et al., 1988; Ellis and Cowan 1999a), mud (Barshaw and Bryant Rich, 1988; personal observation), and salt marsh peat reefs (Able et al., 1988; Barshaw et al., 1994). Suitability of cover may vary with depth. For example, cobble beds are suitable for subtidal settling, but would not be useful intertidally because small round rocks tend to roll in response to disturbances such as wave action. Intertidally, preferred rocks measured approximately 30 x 30 x 10 cm in dimension and substrates found under lobster rocks included silt, mud, sand and shell hash (Cowan, 1999a). Underneath rocks, smaller sediment sizes have greater water retention and may therefore be preferred intertidally. However, many of the lobsters dug depressions where water pooled under their rocks thereby increasing moisture.
Lobsters are likely to be found in intertidal areas where current speeds, salinities, and summer water temperatures are relatively high. Lobster nursery areas may occur as a band from the lower intertidal to shallow depths. In general, densities appear to be higher at depths of 0.4 and 5 m below mean low water than at depths of 10 m (Incze and Wahle, 1991; Wahle and Steneck, 1991; Cowan, 1999a; Wilson, 1999).
Postlarvae may not be expected to settle in deep water due to the distance postlarvae would have to swim from the surface to the bottom. Greater distances from the surface waters to benthic settlement habitats may result in a higher rate of encounter with fish predators(Boudreau et al., 1992; Barshaw and Rich, 1997). The lower intertidal zone may represent an area of enhanced survivorship by acting as a refuge from high predation intensity. Low abundance of large predatory fish characteristic of coastal waters nearshore (Witman and Sebens, 1992) may also play a role in the success of benthic recruitment. Although there is some predation in the intertidal zone, it may be less intense than in the subtidal zone. Bird predators, such as black-backed and herring gulls (Larus sp.), are restricted to feeding on lobsters at certain times of the day only if the tide is low and seas are calm. In contrast, fish predators can actively hunt throughout the tidal cycle depending on their diurnal versus nocturnal activity levels.
Settlement in shallow water may optimize growth rates due to superabundant food supplies and elevated temperatures. Shallow waters tend to be warmer and growth rates in lobster increase proportionally with increased temperature (Waddy et al., 1995). The recapture data gathered using this method may be useful in resolving age cohorts and comparing growth rates and molt increments at various temperatures in the wild.
Temporal Resolution of Intertidal Lobster Monitoring Program
Observations of multiple captures up to three years apart indicate prolonged use of the lower intertidal zone by individuals of H. americanus (Figs. 2 and 3). Most ecological observations of juvenile lobster in the subtidal zone have been restricted to summer months (Hudon, 1987; Able et al., 1988; Heck et al., 1989; Wahle and Incze, 1997; Wahle and Steneck, 1991). Intertidal sampling allows for observations of daily and seasonal patterns of behavior.
A seasonal pattern of abundance was observed such that the highest densities were recorded from May through November. Lower densities in December through April were most likely due to seasonal migrations of yearlings and older juveniles to deeper water since these individuals disappeared and reappeared abruptly. Further evidence for such movements is provided by results of the tagging study. For example, lobster #2 (Figure 4) was captured during three consecutive summer months, disappeared in autumn, and was never captured in winter. Other multiple lobsters were also not typically recaptured in winter. Further analyses of these recaptured lobsters may provide important information on growth rates, survivorship of year classes and indicate patterns of movements in terms of distances traveled over time.
Due to the ease of sampling out of the water, the ILMP method for measuring lobster abundance allows for monthly resampling and thus results in an accurate time series that may be used to develop and test predictive models for annual yields and recruitment. An accurate time series can result because the sampling method is nondestructive - it does not disturb the sediments or remove organisms from the study site. Multiple resamples can also be made subtidally via suction sampling, but it takes a long time for the area sampled to recover because the air lift removes the infaunal organisms as well as lobsters and a good part of the substrate. With repeated samples, the suction-sampled environment could become so degraded that it resembles a desert patch where it is unlikely that postlarval lobster will successfully settle. Therefore, in terms of developing a time series, subtidal suction sampling only yields a "scan" sample at a particular time and then it is necessary to either wait a long time, or sample elsewhere. Sampling by overturning rocks in the lower intertidal zone gives on-going samples, month after month, year after year in the same location.
Potential Basic and Applied Significance of Intertidal Lobster Assessments
In terms of basic ecology, the ILMP may provide a model study for understanding the processes that underlie local population dynamics of a long-lived, large bodied, benthic invertebrate. A full understanding of population dynamics requires knowledge of the rates of birth (recruitment), death, immigration, and emigration. Caley et al. (1996) argue that what is needed to understand the role of recruitment in open populations are multifactorial experiments in which recruitment and postrecruitment processes are simultaneously manipulated. However, before such experiments are appropriate it is necessary to have an indication of baseline levels of settlement and abundances. The ILMP can provide such baseline data for the American lobster.
Taken together the measures of abundance, observations of behavior, and tagging data collected using this method may lead to an accurate time series including numbers of individuals, resolution of age classes, and mortality estimates. Such information is critical in forecasting future stock abundance and detectable changes may serve as an early warning several years before stock collapse. Homarus is characterized as a long-lived genera based on estimates of growth rates and large sizes achieved (specimen weighing over 40 pounds have been recorded). Size at maturity (approximately one pound) is similar to size at recruitment to the fishery. Our current knowledge of growth rates of H. americanus is largely based on laboratory studies. Field studies where hatchery-reared juveniles of H. gammarus were tagged at a few months of age, released into the wild, and subsequently recaptured in harvester's traps seven years later (Bannister et al., 1994) verify age estimates of marketable European lobster. Based on the long time interval between benthic settlement and recruitment to the fishery, an impending stock collapse could go relatively unnoticed for several years in the absence of reliable predictors of sustainable yield.
Life history information is of particular value in the case of H. americanus due to the economic importance of the industry that the species supports throughout the Northwest Atlantic coast of North America. The results of the Intertidal Lobster Monitoring Program indicate that effective protection of the lobster resource may rely not only on fishing regulations, but, also on protecting coastal habitats. The intertidal zone is a particularly vulnerable habitat due to its close proximity to major anthropogenic impacts such as land sources of pollution, fresh water and road runoff, and erosion and sedimentation brought about through poor management practices in coastal development.
Conclusions
The results presented here introduce a new method for monitoring juvenile lobsters where they can be found intertidally. They exemplify the power of using land-based survey methods to assess abundance, time of settlement and growth of juvenile lobsters for the first 4 to 5 years of life. The Intertidal Lobster Monitoring Program takes advantage of lobsters’ use of the lower intertidal zone in order to implement an intensive, long-term, spatially extensive survey of benthic recruitment and growth.
The greatest advantage of the method is the ability to sample at any time of year for long sampling periods regardless of most environmental conditions, including photoperiod and foul weather. Inclement weather can restrict other sampling methods including diving, trawl surveys and trap studies. Intertidal sampling also allows for sampling with minimal disturbance to both the lobster and its habitat. Using this method, lobster are hand-captured with ease, tagged in situ and immediately returned to their shelters. Although the ILMP may be useful as a powerful assessment tool, it is prudent to keep in mind that the intertidal zone represents the most landward margin of lobster distribution and thus may not be representative of the entire population. Therefore, it has been suggested that the Intertidal Lobster Monitoring Program should be used in conjunction with subtidal studies (Cowan 1999a). Recent results indicating a strong correlation between lobster densities in the intertidal and subtidal zones (Figure 7) support the ILMP’s validity as an effective and valid technique for monitoring lobster abundance.
In order to maximize the value of monitoring studies on young lobsters, it is critical that these studies be continued from year to year. Long-term studies are crucial because it takes 6 to 7 years for lobsters to reach minimum legal size. The data collected by the ILMP can help us to gauge the health of the lobster population over time, and may provide a mechanism to detect potential problems long before recruitment of adults to the fishery. Our fisheries-independent research provides pertinent information in a style that is accessible to the public. Through our research we can thereby foster a unique, common-sense approach to fisheries management.
Acknowledgments
This project was supported by the Lobster Advisory Council, Davis Conservation Foundation, Island Institute, Maine Community Foundation, New England Grassroots Environment Fund, Spicewood Fund, and Wildwood Fund. Maine Sea Grant provided funding to purchase monitoring and tagging equipment. We wish to thank the many volunteers who dedicated their time and effort to collecting data.
References
Able, K.W., K.L. Heck, Jr., M.P. Fahay, and C.T. Roman. 1988. Use of salt-marsh peat reefs by small juvenile lobsters on Cape Cod, Massachusetts. Estuaries 11(2): 83-86
Bannister, R.C.A., J.T. Addison, and S.R.J. Lovewell. 1994. Growth, movement, recapture rate and survival of hatchery-reared lobsters (Homarus gammarus Linnaeus, 1758) released into the wild on the English east coast. Crustaceana 67: 156-172.
Barshaw, D.E. and D.R. Bryant-Rich. 1988. A long-term study on the behavior and survival of early juvenile American lobster, Homarus americanus, in three naturalistic substrates: eel grass, mud, and rocks. Fisheries Bulletin, United States 86: 789-796.
Barshaw, D.E, and K.L. Lavalli. 1988. Predation upon postlarval lobsters, Homarus americanus, by cunners, Tauogolabrus adspersus, and mud crabs, Neopanope sayi, on three different substrates: eelgrass, mud, and rocks. Marine Ecology Progress Series 48: 119-123.
Barshaw, D.E. and D.R. Rich. 1997. An analysis of substrate selection by postlarval American lobster, Homarus americanus, using a dynamic optimization model. Oikos 80: 554-564.
Barshaw, D.E, K.W. Able, and K.L. Heck. 1994. Salt marsh peal reefs as protection for postlarval lobsters, Homarus americanus, from fish and crab predators: comparisons with other substrates. Marine Ecology Progress Series 106: 203-206.
Bernstein, B.B. and A. Campbell. 1983. Contribution to the development of methodology for sampling and tagging small juvenile lobsters (Homarus americanus). Canadian Manuscript Report of Fisheries and Aquatic Sciences, Number 1741, pp. 1-34.
Boudreau, B., Y. Simard, and E. Bourget. 1992. Influence of a thermocline on vertical distribution and settlement of post-larvae of the American lobster Homarus americanus Milne-Edwards. Journal of Experimental Marine Biology and Ecology 162: 35-49.
Caley, M.J., M.H. Carr, M.A. Hixon, T.P. Hughes, G.P. Jones, and B.A. Menges. 1996. Recruitment and the local dynamics of open marine populations. Annual Review of Ecology and Systematics 27: 477-500.
Campbell, A. 1991. Juvenile Homarus americanus studies from McNutt Island, Nova Scotia. Abstract. J. Shellfish. Res. 10, 283-284.
Cowan, D.F. 1999a. Method for assessing relative abundance, size-distribution, and growth of recently settled and early juvenile lobster (Homarus americanus) in the lower intertidal zone. Journal of Crustacean Biology, 19(4): 738-751.
Cowan, D.F. 1999b. Intertidal sampling for lobsters. In: Lobster Stock Assessment: Towards Greater Understanding, Collaboration and Improvement. Edited by P.M. Farrey, M.L. Mooney-Sues, and H.C. Tausig. Published by New England Aquarium, Boston. pp. 41-46
Cowan, D.F., J.K. Kanwit, and S.L. Ellis. 1999. Field Handbook: Intertidal Lobster Monitoring Program. Published by The Lobster Conservancy, Friendship, Maine. 48 pp.
Ellis, S.L. and D.F. Cowan. 1999a. Final Report: Intertidal Lobster Monitoring Program: Penobscot Bay Lobster Collaborative, October 1999, submitted to the Island Institute, Rockland, Maine. 27 pp.
Ellis, S.L., and D.F. Cowan 1999b. Preliminary Report on the Intertidal Lobster Monitoring Program, Harpswell, Maine 1998. 8 pp.
Ellis, S.L., and D.F. Cowan 1999c. Preliminary Report on the Intertidal Lobster Monitoring Program, Odiorne Point State Park, New Hampshire 1998. Submitted to New Hampshire Fish and Game Dept., 7 pp.
Heck, K.L., Jr., K.W. Able, M.P. Fahay, and C.T. Roman. 1989. Fishes and decapod crustaceans of Cape Cod eelgrass meadows: species composition, seasonal abundance patterns and comparison with unvegetated substrates. Estuaries 12(2): 59-65.
Hudon, C. 1987. Ecology and growth of postlarval and juvenile lobster, Homarus americanus, off Iles de la Madeleine (Quebec). Can. J. Fish. Aquat. Sci. 44: 1855-1869.
Incze, L.S., and R.A. Wahle. 1991. Recruitment from pelagic to early benthic phase lobsters, Homarus americanus. Marine Ecology Progress Series 79: 77-87.
Krouse, J.S., and G.E. Nutting. 1990. Evaluation of coded microwire tags inserted in legs of small juvenile American lobsters. Amer. Fish. Soc. symp.7: 304-310.
Waddy, S.L., D.E. Aiken, and D.P.V. De Kleijn. 1995. Control of growth and reproduction. In: J. R. Factor, ed., Biology of the lobster, Homarus americanus. Pp. 217-266. Academic Press, New York, New York.
Wahle, P.A., and Incze, L.S. 1997. Pre and post-settlement processes in recruitment of the American lobster. Oikos 63: 1-9.
Wahle, R.A., and R.S. Steneck. 1991. Recruitment habitats and nursery grounds of the American lobster Homarus americanus: a demographic bottleneck? Mar. Ecol. Prog. Ser. 69:231-243.
Wilson, C. 1999. Masters thesis. University of Maine, Orono.
Witman, J.D., and K.P. Sebens. 1992. Regional variation in fish predation intensity: a historical perspective in the Gulf of Maine. Oecologia 90: 305-315.
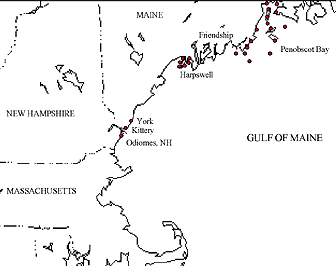
Figure 1. Approximate locations of study sites (solid circles) in The Lobster Conservancy’s Intertidal Lobster Monitoring Program. By autumn 1999, volunteers were monitoring 24 sites in Maine (15 in Penobscot Bay, 1 in Friendship, 6 in Harpswell, 2 in southern Maine), and 1 site in New Hampshire.
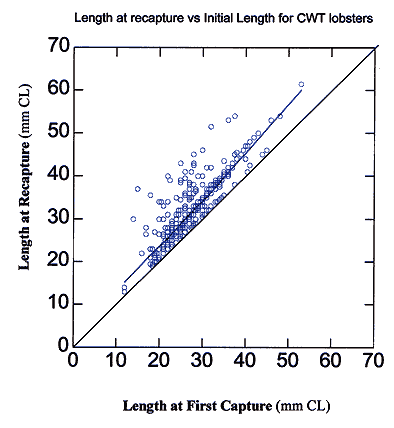
Figure 2. Change in carapace length (mm CL) plotted as CL at first capture versus CL at subsequent capture of the lobsters tagged and recaptured using coded wire tags between September 1993 and December 1999. Each open circle represents one individual. Line drawn through circles indicates best fit for data. Line drawn through origin indicates the line of zero growth.
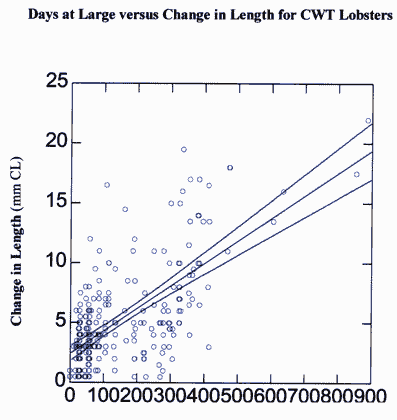
Figure 3. Change in carapace length (mm) over time in number of days for lobsters marked using coded wire tags between September 1993 and December 1999. Lines indicate best fit and 95% confidence interval.
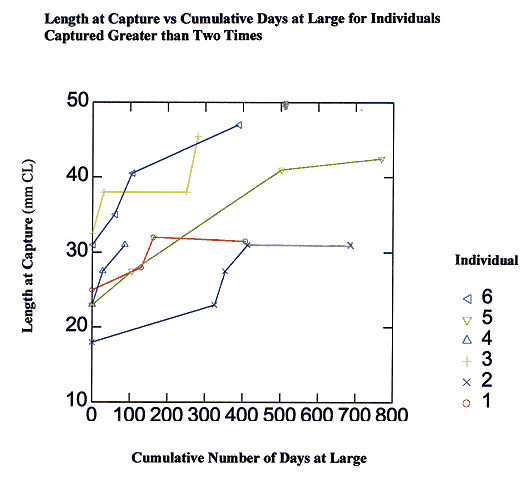
Figure 4. Case histories of six individual juvenile lobsters that were captured more than two times, showing change in carapace length over time relative to number of days at large.
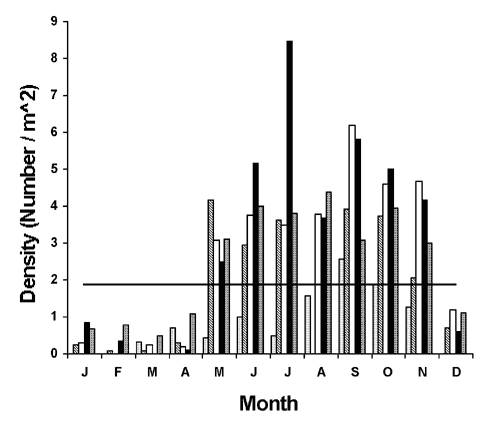
Figure 5. Mean lobster density (number / m2) in the intertidal zone at Lowell's Cove from 1993-1997. Horizontal line through graph represents average density for all five years combined. Dotted bars = 1993; diagonal bars = 1994; open bars = 1995; filled bars = 1996; cross-hatched bars = 1997.
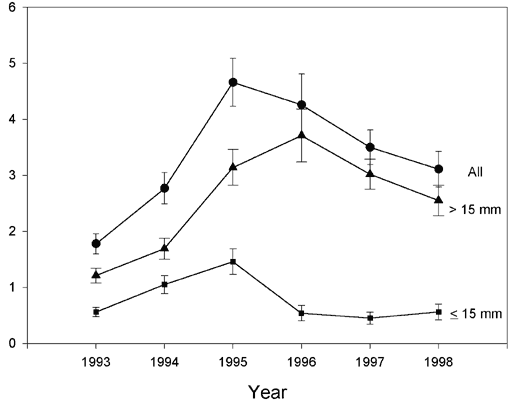
Figure 6. Time series of densities of lobsters found along Transect 1, Lowell's Cove, Casco Bay, Maine from September-November, 1993-1998. The two lower lines represent young-of-the-year (< 15 mm CL) and larger juveniles (> 15 mm CL), while the upper line represents both size classes combined.
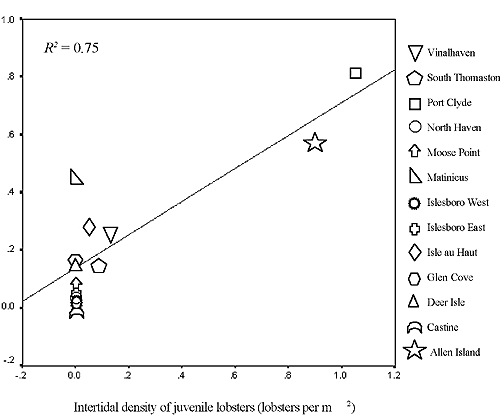
Figure 7. Density of juvenile lobsters (< 40 mm carapace length) as measured at 13 intertidal and subtidal sites around Penobscot Bay, Autumn 1999. Data collected in the intertidal zone (Cowan and Ellis, unpublished) correlates strongly with data collected in the subtidal zone (Steneck, unpublished).
APPENDIX:
Presentations August 1998 — December 1999
| Date |
Speaker |
Event |
Title |
Location |
| 8/10/98 |
D.F. Cowan |
Volunteer Training |
Volunteer Monitoring for Intertidal Lobsters in Penobscot Bay |
South Thomaston, ME |
| 9/3/98 |
D.F. Cowan |
Information Session |
Volunteer Monitoring for Intertidal Lobsters in Penobscot Bay |
Vinalhaven, ME |
| 9/10/98 |
D.F. Cowan |
Volunteer Training |
Volunteer Monitoring for Intertidal Lobsters in Penobscot Bay |
Vinalhaven, ME |
| 9/16/98 |
D.F. Cowan |
Information Session |
Intertidal Sampling for Lobsters |
Colby College, Waterville, ME |
| 9/21/98 |
D.F. Cowan |
Information Session |
Volunteer Monitoring for Intertidal Lobsters in Penobscot Bay |
Deer Isle, ME |
| 9/23/98 |
D.F. Cowan |
Information Session |
Volunteer Monitoring for Intertidal Lobsters in Penobscot Bay |
SUNY Stony Brook, NY |
| 9/24/98 |
D.F. Cowan |
Information Session |
Intertidal Sampling for Lobsters |
Rutger's University, NJ |
| 10/4/98 |
D.F. Cowan |
Volunteer Training |
Volunteer Monitoring for Intertidal Lobsters in Penobscot Bay |
Deer Isle, ME |
| 10/8/98 |
D.F. Cowan |
Information Session |
Volunteer Monitoring for Intertidal Lobsters in Penobscot Bay |
Allen Island, ME |
| 10/9/98 |
D.F. Cowan |
Volunteer Training |
Volunteer Monitoring for Intertidal Lobsters in Penobscot Bay |
Vinalhaven, ME |
| 10/9/98 |
D.F. Cowan |
Information Session |
Intertidal Sampling for Lobsters |
Maine Coastal Program: Sea & Shore Conference, Rockport, ME |
| 11/17/98 |
D.F. Cowan |
Scientific meeting |
Intertidal Monitoring for the Pen Bay Lobster Collaborative |
Camden, ME |
| 3/5/99 |
D.F. Cowan |
Conference presentation |
Intertidal Sampling for Lobsters |
U.S./Canadian Lobster Summit III, Rockport, ME |
| 3/18/99 |
S.L. Ellis |
Round Table |
Volunteer Monitoring for Intertidal Lobsters in Penobscot Bay |
Vinalhaven, ME |
| 3/26/99 |
S.L. Ellis |
Radio show |
Penobscot Bay Collaborative Research Project: Volunteer Monitoring |
WERU 89.9 FM, "Talk of the Towns"
Blue Hill, , ME |
| Date |
Speaker |
Event |
Title |
Location |
| 4/19/99 |
S.L. Ellis |
Volunteer Training |
Volunteer Monitoring for Intertidal Lobsters in Penobscot Bay |
Isle au Haut, ME |
| 5/15/99 |
S.L. Ellis |
Information Session |
Penobscot Bay Collaborative Research Project: Intertidal Monitoring |
Islesboro, ME |
| 5/16/99 |
S.L. Ellis |
Volunteer Training |
Volunteer Monitoring for Intertidal Lobsters in Penobscot Bay |
Islesboro, ME |
| 5/17/99 |
S.L. Ellis |
Round Table |
Penobscot Bay Collaborative Research Project: Intertidal Monitoring |
Rockland, ME |
| 5/18/99 |
S.L. Ellis |
Volunteer Training |
Volunteer Monitoring for Intertidal Lobsters in Penobscot Bay |
South Thomaston, ME |
| 5/18/99 |
S.L. Ellis |
Information Session |
Penobscot Bay Collaborative Research Project: Intertidal Monitoring |
North Haven, ME |
| 5/19/99 |
S.L. Ellis |
Volunteer Training |
Volunteer Monitoring for Intertidal Lobsters in Penobscot Bay |
North Haven, ME |
| 5/19/99 |
D.F. Cowan |
Education session |
Life and Sex in the Intertidal Zone |
Pen Bay Marine Volunteers: Searsport, ME |
| 6/14/99 |
S.L. Ellis |
Volunteer Training |
Volunteer Monitoring for Intertidal Lobsters in Penobscot Bay |
Rockport, ME |
| 6/14/99 |
S.L. Ellis |
Volunteer Training |
Volunteer Monitoring for Intertidal Lobsters in Penobscot Bay |
Deer Isle, ME |
| 6/16/99 |
S.L. Ellis |
Volunteer Training |
Volunteer Monitoring for Intertidal Lobsters in Pen Bay |
Monhegan Island, ME |
| 6/17/99 |
S.L. Ellis |
Information Session |
Penobscot Bay Collaborative Research Project: Intertidal Monitoring |
Matinicus Island, ME |
| 6/18/99 |
S.L. Ellis |
Volunteer Training |
Volunteer Monitoring for Intertidal Lobsters in Penobscot Bay |
Matinicus Island, ME |
| 12/8/99 |
S.L. Ellis |
Scientific meeting |
Intertidal Lobster Monitoring in Penobscot Bay, 1998/99 |
Pen Bay Lobster Collaborative, Camden, ME |
| 12/8/99 |
A. Watson |
Outreach |
A Volunteer's Perspective on The Lobster Conservancy's Monitoring Program |
Friends of Casco Bay, Freeport, ME |
Communication of Results to Zone Councils:
To communicate results of this research to Zone Councils, copies of this report were sent in April 2000 to the Chairman of each Zone Council, as listed below:
Zone A:Norbert Lemieux
P.O. Box 325
Cutler, ME 04626
Zone B:Jon Carter
333 Main St.
Bar Harbor, ME 04069
Zone C:Gerald Weed
RFD 1, Box 1764
Stonington, ME 04681
Zone D: David Black
186 Northport Ave.
Belfast, ME 04915
Zone E:Larry Knapp
P.O. Box 153
Boothbay, ME 04537
Zone F:John Bear, Jr.
1517 Harpswell Islands Rd.
Orr's Island, ME 04066
Zone G:James Alwin
32 Old Cape Rd.
Kennebunkport, ME 04046
FINANCIAL REPORT
Expenditures of Lobster Advisory Council Contract # 99872 to date:
| Category |
Cost |
| Research equipment |
535.28 |
| Research supplies |
923.14 |
| Educational supplies |
244.45 |
| Outreach |
772.86 |
| Telephone |
325.84 |
| Mileage |
797.73 |
| Salary |
2,200.78 |
| Total |
5,800.08 |
Planned allocation of remaining $1,450 due from Contract # 99872:
reimbursement for mileage to study sites in 1999
In-kind contributions to tagging program.
| Source |
Item |
Value |
| Maine/NH Sea Grant |
Microwire tags |
$1,100 |
| Lobster Institute, U. Maine |
Microwire tags |
$1,100 |
| Total |
|
$2,200 |







